|
3/10/2024 0 Comments Spinal cord stimulation by abbott
feature the company's proprietary, low-energy BurstDR waveform. All of Abbott's SCS therapies in the U.S. This new indication will span across the entirety of Abbott's SCS portfolio in the U.S., which includes the recharge-free Proclaim SCS family and the rechargeable Eterna SCS platform. Results at six months for the first 200 patients demonstrated clear clinical evidence supporting BurstDR stimulation therapy as an effective treatment for chronic back pain and were sustained at the 12-month follow-up. This labeling expansion was supported by results from the DISTINCT study, which demonstrated that Abbott's proprietary BurstDR SCS technology improved pain levels, ability to perform daily activities and emotional well-being in people with chronic back pain.Ībbott's DISTINCT study, which enrolled 270 participants who suffered for an average of 12.8 years with pain, is the largest randomized controlled trial for SCS in people with chronic back pain when surgery is not an option. Food and Drug Administration (FDA) has approved its spinal cord stimulation (SCS) devices for the treatment of chronic back pain in people who have not had or are not eligible to receive back surgery, known as non-surgical back pain. See product advisories.(RTTNews) - Abbott Laboratories (ABT) announced Tuesday that the U.S. The inability to exit MRI mode would require device replacement surgery to restore therapy. Note: For Proclaim™ family of devices, performing any of the above actions while your device is in MRI mode may result in the inability to exit MRI mode. DO NOT: update, install, or delete the Patient Controller app or Bluetooth ‡ connection.DO NOT: alter, damage, update or lose your Patient Controller handheld.Set your system to MRI mode. This turns off stimulation and protects your device during your MRI scan.If applicable, fully charge your generator.Refer to your user guide for charging instructions. Disable automatic updates for the Patient Controller app and iOS ‡ software before placing the IPG in MRI mode.For Proclaim™ family of devices, before attempting to place your device into MRI mode, and with the help of your physician or Abbott representative, ensure your Patient Controller app is updated to the latest “ Patient Controller NR - US” application from the Apple ‡ App Store ‡.Please see guidance currently available in your Patient Manual. 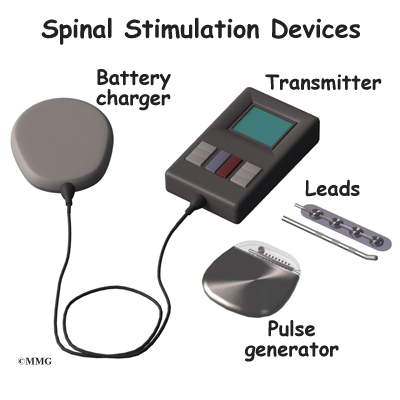
The Abbott devices shown in the table are eligible for an MRI scan within approved parameters.Īlways ask your pain physician to confirm whether your device is eligible for an MRI.ĭepending on your device and which leads have been implanted with your system, you may qualify for an MRI scan of either your whole body or just your head and extremities (arms and legs). Your system may be designed to let you safely receive an MRI scan under certain conditions. Our table can help you identify which kinds of scans are approved for each of our MR Conditional implanted devices. If you have an implanted device, you need to make sure your device is approved to receive an MRI.įortunately, with certain Abbott neurostimulators, you can safely have an MRI scan when conditions for safe scanning are met. But in rare cases, they can interfere with your neurostimulator device, possibly leading to injury. In general, MRI scans are safe, routine procedures. It may be an option if you suffer chronic back, leg or arm pain and have not. It helps people better manage their chronic pain and reduce their use of opioid medications. A small device, similar to a pacemaker, delivers electrical pulses to the spinal cord. 
An MRI uses strong magnets to create images of the organs and tissues in the body. Spinal cord stimulation therapy masks pain signals before they reach the brain.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
